The compounds of the alkali metals are common in nature and daily life. Group 1: The Alkali Metals – lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr) are soft, shiny, and highly reactive metals.For example, the elements of Group 1 are known as the alkali metals, Group 2 are the alkaline earth metals, Group 17 are the halogens, and Group 18 are the noble gases. Chemists often make general statements about the properties of the elements in a group using descriptive names with historical origins. The periodic table is arranged so that elements with similar chemical behaviors are in the same group. A very basic overview of atomic structure. The style attribute specifies the style to give the columns. The span attribute specifies how many columns that get the style. Each group is specified with a element. The element should be used as a container for the column specifications. 1 Find the US States - No Outlines Minefield 2 10 Closest Capitals to Brussels 3 Pick the NBA Logos 4 Countries of the World Science Elements Random Science or Elements Quiz Find the Element Groups Can you click the element groups of the periodic table By teedslaststand - /5 - RATE QUIZ MORE INFO Picture Click Forced Order Best Score Go Orange. Element Games Games based on the Table of Elements Featuring: Element Flash Cards Element Crossword Puzzles Element Math Game Element Concentration Element Hangman Element Matching. If you want to style the two first columns of a table, use the and elements. Element Games Games based on the Table of Elements Featuring: Element Flash Cards Element Crossword Puzzles Element Math Game Element Concentration Element Hangman Element Matching. If you want to style the two first columns of a table, use the and elements.
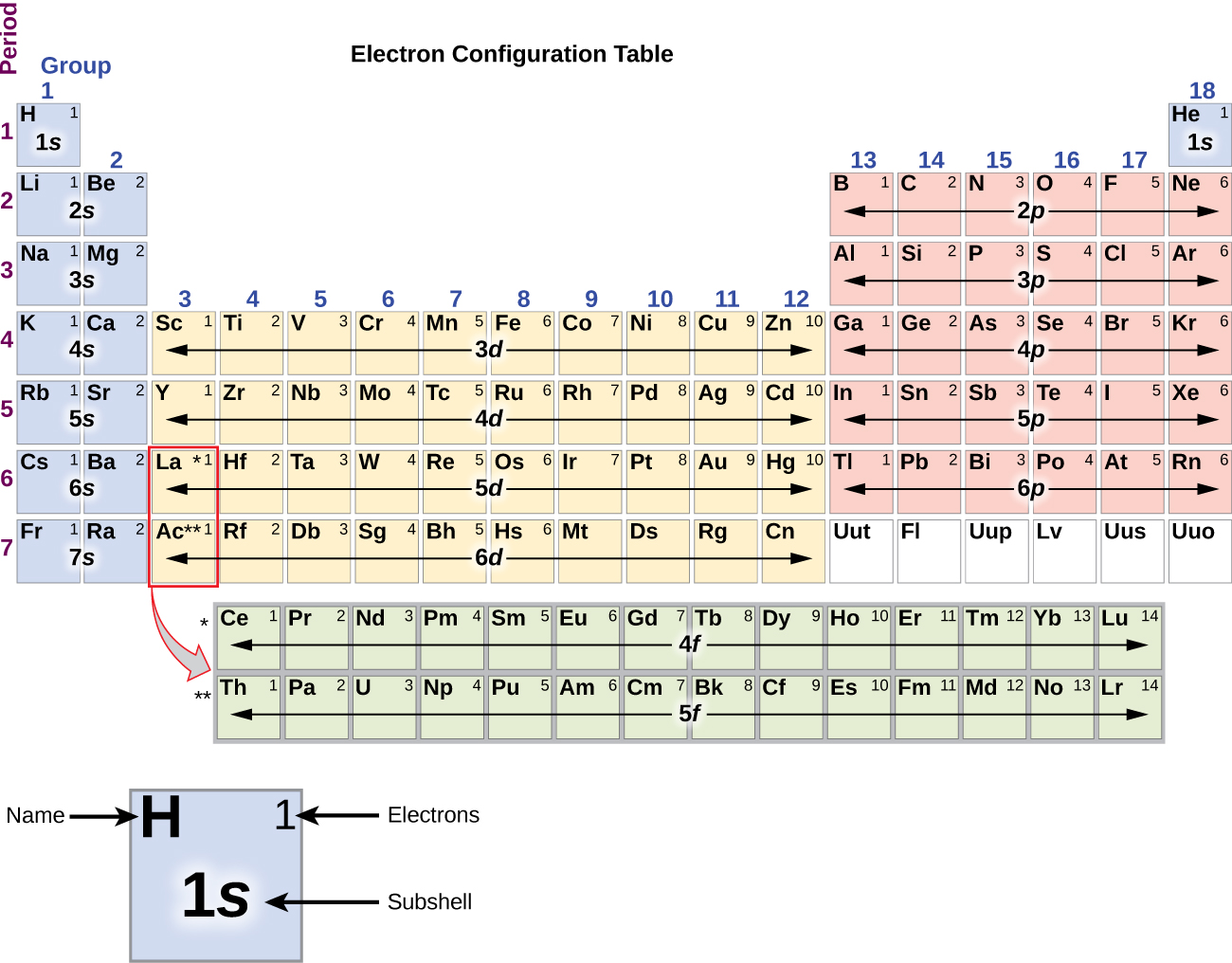
The trends of the atomic radii and melting points (and other chemical and physical properties of the elements) can be explained by the electron shell theory of the atom discussed in the following sections. An easy to understand Table of Elements listing basic physical and historical data. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
